
The Prelude on Novel Receptor and Ligand Targets Involved in the Treatment of Diabetes Mellitus
Abstract
Metabolic disorders are a group of disorders, due to the disruption of the normal metabolic process at a cellular level. Diabetes Mellitus and Tyrosinaemia are the majorly reported metabolic disorders. Among them, Diabetes Mellitus is a one of the leading metabolic syndrome, affecting 5 to 7 % of the population worldwide and mainly characterised by elevated levels of glucose and is associated with two types of physiological event disturbances such as impaired insulin secretion and insulin resistance. Up to now, various treatment strategies are like insulin, alphaglucosidase inhibitors, biguanides, incretins were being followed. Concurrently, various novel therapeutic strategies are required to advance the therapy of Diabetes mellitus. For the last few decades, there has been an extensive research in understanding the metabolic pathways involved in Diabetes Mellitus at the cellular level and having the profound knowledge on cell-growth, cell-cycle, and apoptosis at a molecular level provides new targets for the treatment of Diabetes Mellitus. Receptor signalling has been involved in these mechanisms, to translate the information coming from outside. To understand the various receptors involved in these pathways, we must have a sound knowledge on receptors and ligands involved in it. This review mainly summarises the receptors and ligands which are involved the Diabetes Mellitus. Finally, researchers have to develop the alternative chemical moieties that retain their affinity to receptors and efficacy. Diabetes Mellitus being a metabolic disorder due to the glucose surfeit, demands the need for regular exercise along with dietary changes.
Introduction
The human body has eleven systems, namely central nervous, cardiovascular, endocrine system etc. Among all, the endocrine system works in a distinct fashion i.e. away from the synthesis area. To function normally in every aspect of cellular pathway, there is a need for the cells to be in a harmonised state. Every cellular part has different sensors, namely receptors. Receptors are dynamic protein structures, in inactive mode at the cell surface or in the cytoplasm or in nucleus, activated by ligands to receive the chemical signals from outside. Ligands are the binding molecules that mediate their action through their receptors. Ligands are mainly of two types i.e. agonists and antagonists. The cellular response depends on the type of molecule which binds to the receptor. In the present days, vast research being carried out on the cellular pathways involves the reaction between the receptor and the ligand. This, in turn provides new pathways to discover more selective therapeutic drugs.
Diabetes Mellitus (DM) is a chronic multifactorial metabolic disorder resulted due to the altered homeostasis between glucose production and its metabolism.1 DM is mainly characterised by hyperglycaemia, altered lipid metabolism ascribed due to the unsubstantial amount of insulin production by the β-cells, secretion or both and insulin resistance, oxidative stress, and inflammation.2DM is a 5th leading cause for mortality, prevalence of DM in adults was 285 million (6.4%) in 2010, and this value is predicted to reach around 439 million (7.7%) by 2030. DM is mainly of two types i.e., Type-1 or Insulin dependent DM (IDDM) and Type 2 or non-insulin dependent DM (NIDDM).3 Type 2 DM is accounts for at least 90% of cases and it is the predominant form.4 Up to now, various types of treatments are used in the management of DM based on different types of pharmacological actions, but these drugs aren’t able to mitigate the disease progress, although the insulin resistance is an unravel mechanism for scientists. Moreover, DM is associated with various microvascular (retinopathy, nephropathy, and neuropathy) and macrovascular (coronary, cerebral, and peripheral) complications, which necessitated further extensive multidisciplinary research aimed to treat the DM.
The main pathological event in the DM is insulin resistance (IR). Generally IR is defined as decreased insulin action to activate downstream signalling at a cellular level to normalize the glucose surfeit.5 The major proposed mechanism for the negative insulin signalling was phosphorylation of serine residues like IRS-1, which includes ser636, ser312, and ser1101. In the phosphorylated state, IRS-1 impedes interaction with insulin receptor signalling and ultimately results in insulin resistance.6
In this review, we have summarised, a detail description of receptors and ligands of the DM which includes glucose-transporters, calcium-sensing receptors (CaR), epidermal-growth factor(EGF), estrogen-receptor(ER), farnesoid-X-receptor(FXR), gastrin-releasing peptide(GRP), lyso-phosphatidic acid, muscarinic-receptor, neurotensin, peroxisome- proliferator activated receptor (PPAR), insulin receptor, adiponectin receptor, adenosine receptor, cholecystokinin(CCK), cannabinoid receptor(CB), Insulin-like growth factor(IGF), neuropeptide Y(NPY),toll-like receptor(TLR), and protein-tyrosine phosphatase (PTPs)-1B inhibitors.
In this review we underscore the quick glance over the receptors and ligands involved in the DM, and their interaction in physiological, pathological events in the DM.
Glucose transporters (GLUT)
Glucose is a major metabolic fuel for all living mammalian cells, which is required for their normal physiological functions. Generally, glucose was transported across the cell-membrane by the two different mechanisms i.e sodium-independent (facilitated diffusion, GLUT transporters) and sodium-dependent (secondary active transport, SGLT transporters) with varied kinetic properties. Glucose transporters (GLUT) are available in 12 different isomeric forms belonging to SLC2A hexose transport family, and six different SGLT are available belonging to SLC5A co-transport family. Among them all GLUT4 is mainly involved in glucose transport across cell-membrane of brown and white adipose tissue, muscle (skeletal), heart (myocardium)7, owing to which, a mutation in GLUT4 causes the DM.8 Almost, 90% of the filtered glucose was reabsorbed by the sodium-glucose cotransporter-2, at the S1 segment of the proximal tubule, remaining is absorbed by the SGLT1 at the S3 segment.9 The maximum level of reabsorption was observed in the Type-1 Diabetic patients, implying one of the cause of diabetic condition. The increased levels of SGLT2 was observed in alloxan-induced diabetic rats,10 and GLUT2 levels were increased in streptozotocin diabetic rats.11 The inhibition of SGLT transporters improves diabetic conditions in streptozotocin induced diabetic rats.12 By inhibiting the SGLT2 receptors, the levels of plasma glucose were reduced significantly.13 Indeed, SGLT2 receptor antagonists are the promising approach to treat DM.
Calcium Sensing Receptors (CaR)
Calcium sensing receptors plays a major role in regulation of pancreatic β-cells’ insulin secretion to maintain the glucose homeostasis, and also involve in β-cell development, and growth. Levels of calcium sensing receptors are variable qualitatively or quantitatively in diabetic animal models. Extracellular calcium is involved in insulin secretion, followed by concentration dependent, reversible inhibition of secretion. The mechanism involved in that is increase in cyclic AMP levels, through the phospholipase C–IP3pathway. CaR are mainly stimulated by the cations like Ca2+, Mg2+ and amino acids like L-Phe in HEK-293 cell lines. Apart from that, these are activated by the calcimimetic agents like phenylalkylamines, NPS R-467 and NPS R-568.14 Elizabeth Gray et al. observed that calcimimetic R-568 agonist activates the concentration of extracellular Ca2+ and subsequently activates CaR receptors and induces the insulin release from human islets and MIN6 cells. The mechanism involved in CaR mediated activation is associated with p42/44 mitogen-activated protein kinases (MAPK), and its activation is inhibited by the p42/44 inhibitors. Moreover, CaR mediated insulin secretion is decreased by inhibitors of Phospholiase c, calcium-calmodulin dependent kinase inhibitors and also there is involvement of protein-kinase C inhibitors. Thus, Calcium is involved in exocytosis of insulin secretion.15 Thus, calcium sensing becomes effective in the treatment of DM.16
Epidermal Growth Factor (EGF)
Development and progression of DM is associated with the EGF-receptor ligand system. EGF receptor is an 1186 amino acid glycoprotein, single transmembrane tyrosine-kinase type of receptor, mainly involved in the development of microvessel myogenic tone nature and cell processes like proliferation, survival, and differentiation during development, tissue homeostasis. Leptin level in DM increases the EGFR tyrosine kinase phosphorylation, leading to activation of ERK1/2 MAP-kinase. However, increasing the activity of EGFR phosphorylation leads to increase in glucose levels.17,18 And, EGFR ligand like betacellulin is associated with the increase in β-cell proliferation and its neogenesis and increase in the activity of gut hormone like GLP-1.19 This provides new direction to discover the new molecular targets in DM.
Estrogen Receptor(ER)
Estrogen is having the effect on vascular system and DM.20 The prevalence of DM is more in men, when compared to women,21explains the role of steroid hormone estrogen in the protective ability against the disease progress. ER receptors are of two types namely ERα and ERβ. Estrogen is mainly involved in the regulation of glucose homeostasis. ERβ agonists also helpful in DM,22 ERα may interfere with the one of the inflammatory pathway i.e cytokine-driven iNOS pathway in DM hyperglycaemic rats.23 This warrants, selective ER agonists are useful in DM along with further extensive research in this area.
Farnesoid X Receptor (FxR)
Farnesoid X receptor (FXR) is a nuclear receptor involved in hepatic glucose and lipid metabolism is mainly found in the intestine, kidneys, and adrenal glands. It is widely expressed in the gastro-intestinal tract. In the last few years the Farnesoid receptor activity has been reviewed.24 Bile acids (BA) play an important role in Farnesoid receptor activation through gluconeogenesis.25FxR activates the induction of the glucose regulated transcription factor KLF11, and FxR receptor activation in beta TC6 cells increases the AkT phosphorylation subsequently causing translocation of GLUT2 transporters at cellular membrane. In another, experiment in non obese diabetic (NOD) mice activation of FxR receptor delay the development of signs and symptoms of hyperglycaemia, diabetes, and glycosuria.26 In another experiment FxR activation led to the reduced expression of glucogenic enzymes like G6Pase (glucose-6-phosphatase), PEPCK (phosphoenol pyruvate kinase), and FBP1 (fructose-1,6-bisphosphatase).27,28 FxR receptor activation by the synthetic agonist like GW4064 in insulin-resistant ob/ob mice reduced hyperinsulinemia and improved glucose tolerance.29 These results suggest that FxR agonists are helpful in the treatment of DM in future.
Gastrin releasing peptide (GRP) Receptor
Gastrin releasing peptide (GRP) is a 27 amino acid, neuropeptide strongly present in the gastro-intestinal tract mainly involved in the digestion, and metabolism. GRP primarily activates the insulin secretion in-vitro and in-vivo by both direct activation of islet cells and indirect activation of ganglionic neurons through gastrin releasing peptide receptor (GRPR).30 GRP stimulation in L cells of intestine activates the mitogen activated protein kinase (MAPKK) and subsequent phosphorylation of p44/42 mitogen activated protein kinase (MAPK). In another, GRP mediated stimulation activates the cholecystokinin through protein kinase C (PKC).31Inactivation of PKCs by the phorbol myristate prevents the insulin secretion from β-cells which is mediated by the GRPs.32
Lyso-phosphatidic acid (LPA)
Lysophosphatidic acid (LPA) is a potent arbitatory agent that mediates mainly smooth muscle contraction, platelet aggregation, anti apoptosis, cell rounding, and cell proliferation regulation.33LPA mainly present in the blood, which is produced by lysophospholipase D (Lyso PLD) enzyme.34 LPA was found to enhance the glucose uptake in 3T3-L1 adipocytes and GLUT4myc myotubes by triggering GLUT4 translocation to the plasma membrane. Although, if the effect of LPA on glucose uptake was inhibited by the LPA antagonists like Ki16425 and Gi inhibitor pertussis toxin, LPA showed the blood glucose lowering effect in streptozotocin induced DM.35 So, LPA acts as a potent modulator of glucose homeostasis in adipose and muscle tissues.
Muscarinic receptors
Muscarinic receptors are G-protein coupled receptors (GPCR) involves in calcium mobilisation, phospholipase-c and protein kinase-c activation. In human beings, the known endogenous cholinergic agonists are acetylcholine (Ach), and conjugated secondary bile acids (BAs).36 CHRM3 are located in many places in the body, e.g. smooth muscle, the endocrine glands, exocrine glands, pancreas. Elevated levels of CHRM3 mainly expressed in pancreatic B-cells, and muscarinic receptors activate the GLP-1 peptide secretion by the indirect mechanism involving vagus nerve and from the from the proximal intestine L cells.37
Muscarinic receptor signalling in Insulin secretion
Muscarinic receptor ligands, such as secondary BAs and Ach stimulate the extracellular muscarinic receptors. The main mechanism involved in insulin release is through G-protein coupled receptor signalling to the calcium and PKC pathways. The activation of M3 receptor induces the hydrolysis of membrane component phospholipid phosphatidyl inositol- 4,5-biphosphate (PIP2), catalyzed by phospholipase C (PLC). This reaction generates the formation of two secondary messengers i.e. inositol-1,4,5- triphosphate (IP3) and diacylglycerol (DAG). IP3 in turn drives the calcium release from the IP3 sensitive stores, simultaneously DAG activates PKC.38
However, G-protein-independent pathway is also involved in the insulin secretion through the protein-kinase D1(PKD1). The principle involved is, phosphorylated form of M3 activates the G-protein-independent pathway through β-arrestin dependent process resulting in secretory releasing.39 In addition, it activates the sodium channel designated as NALCN, a non-selective sodium-leak channel which plays an important role in insulin release. M3R has showed to activate this channel in the model of MIN-6, pancreatic β cell line via the Src family of tyrosine kinases (SFKs).40 In particular, there is a need of extensive research studies on this mechanism to develop better therapeutic drugs.
Neurotensin (NT)
Neurotensin (NT) is a peptide neurohormone or neuromodulator in the central nervous system and peripheral nervous system.41Neurotensin receptors are present throughout the gut, to express their activity. Neurotensin is involved in different variety of functions including stimulation of pancreatic secretions, stimulation of colonic motility and biliary secretions.42Neurotensin receptors are G-protein coupled type of receptors, i.e NTSR1, NTSR2, NTSR3. NTSR2/NTSR3 involved in the pancreatic β-receptor signalling to mediate the insulin secretion. The mechanism involved in this secretion is that the NT binds to the NTSR2/NTSR3 complex leading to the activation of downstream signalling mechanism and causes activation of phospholipase C which enhances the release of calcium, responsible for insulin secretion. NTSR2 selective agonist, levocabastine, causes a transient increase in intracellular calcium levels in Ins1-E cell line.43 This suggests that refinement of NT can serve as a better novel therapeutic approach for further investigation.
Peroxisome proliferator-activated receptor (PPAR)
PPARs come under the members of nuclear hormone receptor super family and ligand-activated transcription factors. PPARs comprise of mainly three different isoforms: PPARα, PPARβ/γ and PPARγ.44 Activation of PPARα possesses an anti-diabetic effect. PPARγ agonists were used as therapeutic agents for treatment of DM. PPARγ directly activates the GLUT-2 and B-glucokinase in liver.45 Till now, the different well known mechanisms have been reported in the antidiabetic activity of PPARγ agonists, like thiazolidines (TZD), they increase the expression of insulin receptor substrate (IRS)-1,46 IRS-2,47 the cbl-associated protein,48and the p85 subunit of phosphatidyl inositol.49
Insulin Receptor (IR)
Insulin receptor is a tyrosine-kinase mediated heterotetrameric membrane glycoprotein receptor comprising of two α and two β subunits linked by disulphide linkage involved in pleiotropic actions of insulin. The mechanism involved in the insulin receptor signalling is, insulin binds to the extra cellular subunit, bringing the two 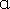 subunits to come together. By this conformational change ATP binds to the β subunit intracellularly and causes autophosphorylation and enables its kinase activity, leading to IRS/PK-I3 pathway and starts the PIP3 dependent kinases.50,51Along with this, another pathway involved in this is exchange factors SOS and growth factor receptor binding protein 2 (GRB2) for stimulation of insulin mediated actions on growth and proliferation.52 Insulin Receptor substrates are insulin, IGF-1,and other cytokine receptors that phosphorylate the specific Y-x-x-M motifs.53 This explains the potential diversity of insulin action.
subunits to come together. By this conformational change ATP binds to the β subunit intracellularly and causes autophosphorylation and enables its kinase activity, leading to IRS/PK-I3 pathway and starts the PIP3 dependent kinases.50,51Along with this, another pathway involved in this is exchange factors SOS and growth factor receptor binding protein 2 (GRB2) for stimulation of insulin mediated actions on growth and proliferation.52 Insulin Receptor substrates are insulin, IGF-1,and other cytokine receptors that phosphorylate the specific Y-x-x-M motifs.53 This explains the potential diversity of insulin action.
Adiponectin Receptors
Adipose tissue produces different types of small bioactive molecules, i.e adipocytokines54 also called adipocyte complement-related protein of 30 kDa (Acrp30).55 Obesity is a principal cause of cardiovascular disease, and DM. Adiponectin receptors are found to be available in two isomeric forms viz. Adiponectin Receptor R1 (Adipo R1) and Adiponectin Receptor R2 (Adipo R1 is abundantly expressed in muscle, and Adipo R2 is mainly present in liver). Adiponectin mediates its action through these receptors via intracellular signalling pathway by using AMP-activated protein Kinase (AMPK), Peroxisome-proliferated activated receptor-α (PPAR-α), fatty-acid oxidation and glucose uptake in liver. Adiponectin levels were measured by the Enzyme-linked immunosorbent Assay (ELISA), to correlate the adiponectin and DM.56 Adiponectin levels were decreased in DM type-2 i.e inversely related to glucose levels. But, excitingly the levels of adiponectin were increased in type-1 DM i.e positively correlated with insulin sensitivity.57 In-case of anti-diabetic drugs increased serum adiponectin levels in insulin resistant cases,58 and increased levels of adiponectin decrease the risk for DM.59 Further more extensive research work needs to elucidate the detailed information.
Adenosine Receptor
Adenosine is a purine metabolic product, one of the potent endogenous autocrine immunosuppressive and anti-inflammatory molecules, released into the extracellular space at the time of tissue injury and inflammation. Adenosine receptors are different types like A1, A2A, A2B, and A3 belongs to G-protein coupled receptors.60 All four receptors are involved in adaptive immunity response by adenosine receptor activation. Moreover, single cup of tea or coffee suffices for the blockade of A1, A2A, A2B receptors, mainly, A2B adenosine receptor (A2bAR) involved in regulation of inflammation. A2B receptor modulated activity in DM was performed by Hillary Johnston-Cox. Up-regulation of A2BAR receptor in control mice along with A2AR knock-out mice, the hallmarks of DM were observed in knock-out mice. Mechanism involved in that A2bAR regulation of SREBP-1 expression, a repressor of insulin-receptor signalling-2(IRS-2), and also A2BAR ligand was observed for 28 days after the high fat, high cholesterol diet (HFD) restored the IRS-2 levels and subsequently abrogated the development of T2D. A1AR receptors were expressed in adipose tissue, principally involved in the dyslipidaemia, insulin resistance, and diabetes. Selective A2AR agonists are helpful in DM by inhibiting the cAMP through G-protein coupled adenylyl cyclase using N6-cyclopentyladenosine.61 However, there is a need for the discovery of adenosine receptor agonists.
Cholecystokinin (CCK)
Cholecystokinin (CCK)62 and its analogue cerulean63 leads to the pancreatic cell growth. CCKA receptors were involved in regulation of pancreatic cell growth and to stimulate the secretion of the digestive enzyme pancreatic lipase, this response was blocked by simultaneous administration of CCKA receptor antagonists64 or CCKA deficient mice.65 Cholecystokinin acts through the G-protein coupled mechanism, like Gq along with phosphoinositide-specific phospholipase C (PLC-β) and thereby elevates intracellular calcium. And also, CCK activates the NFAT pathway through the calcineurin pathway, in a dose-dependent manner this effect was blocked by inhibitors of this phosphatase, cyclosporine A and immune suppressants FK506.66
Cannabinoid Receptor (CB)
Cannabinoid receptors are of two types, i.e. 7 transmembrane CB1 and CB2, belong to Vanilloid-type-1 receptors.CB1 receptors are mainly expressed in the central nervous system, where as CB2 receptors are available in haempoietic system and immune system, mainly regulating the immunological activities.67 Cannabinoid receptors are partly responsible for the activities of natural constituents of Cannabis sativa. CB respond functionally and biochemically to two natural cannabidiol and Δ9tetrahydrocannabivarin. Oxidative stress is involved in different pathological events ranging from pain, cancer, obesity, inflammation, and metabolic disorders especially diabetes and its vascular complications.68 Most of the diabetic complications are due to reactive oxygen species or reactive nitrogen species. Activation of CB1 receptors causes increased insulin secretion. Cannabinoids also activates one of the nuclear receptors peroxisome-proliferated activated receptor-α (PPAR-α). Endocannabinoids like virodhamine, anandamide, and noladin activate the PPAR-α.69 But, conversely CB1 receptor antagonist rimonabant, showed insulin resistance, C-reactive protein and reduced the glycated haemoglobin (Hb1c) levels in insulin treated diabetic patients, in drug naïve and metformin and sulphonylurea treated type 2 diabetic patients.70 CB receptors having the modulatory activity in the DM warrants further extensive research in this area.
Insulin like growth factor (IGF) Receptor
Insulin like growth factor (IGF) has two types of ligands, namely IGF-1 and IGF-2. IGF-1 receptor is a tyrosine-kinase membrane associated receptor, existed as a heterotetramer with two- α and two-β subunits linked through the disulphide linkage. Binding of IGF molecules promotes the intramolecular autophosphorylation and phosphorylation of its critical targets. Moreover, it also activates several other signalling pathways such as P13k/AKT pathway and Ras/MAPK.71 IGF receptors are abundantly present in skeletal muscle, where as adipose, hepatic tissues have fewer. IGF-1 is structurally related to insulin. IGF-1 showed the positive effects like β-cell growth, survival, and insulin secretion.72 Along with these effects, it also produces increased glucose transport in skeletal, adipose tissue, increased glycogenesis, lipogenesis, and decreased lipolysis. It also decreases the blood glucose, free fatty acids levels similar to insulin.73 Thus, targeting the IGF pathway provides an efficient strategy to treat the DM.
Neuro Peptide (NP)Y Receptor
Peptide YY and NPY belonging the family of pancreatic polypeptide hormones, comprise a chain of length 36 amino acids, secreted from the ileal L cells after taking the meals. They posses N-terminal having tyrosine moiety and C-terminal having the amide residue of tyrosine.74 PYY is mostly confined to the endocrine cells of the gut. NPY and Y2 are involved in different activities like appetite and feeding, angiogenesis, carcinogenesis, and DM. NPYergic neurons are inhibitory autoreceptor, can regulate the expression and secretion of NPY and other related neurotransmitters. They are also involved in leptin receptor expression that enables the maintenance of energy homeostasis. Moreover, Y2 receptor deletion in ob/ob mice attenuates the type 2 DM in mice.75
Toll-like Receptors (TLR)
Inflammation and its mediators are involved in different types of diseases like atherosclerosis, cancer, and DM.76 DM is mainly associated with the inflammatory mediators like sialic acid, C-reactive protein, alpha-1 acid glycoprotein etc., suggesting the lack of innate-immunity response.77 TLRs are expressed specifically at the site of tissue injury and interact with endogenous ligands like HSPs (heat-shock proteins) 60/70, oxLDL (oxidised LDL), fibronectin and fibrinogen which are abundantly seen in DM.78–83TLR family consists of 13 different types mammalian receptors. Among them TLR2 and TLR4 are mainly involved in the pathogenesis of insulin resistance (IR), and DM in experimental and clinical conditions.76 Kim HS et al. found that TLR-2 senses the β-cell death as a mediator of inflammation,83 and activation of TLR-2 and TLR-4 leads to the downstream cytokine, p38 MAPK and NF-kB production.76,84,85 In addition to this Song et al.86showed increase in TLR4mRNA expression in differentiating adipose tissue of db/db mice. TLR-2 is involved in streptozotocin induced inflammation through the MyD88-dependent pathway, and TLR-4 also involved in MyD88-independent pathway along with NF-kB pathway.87 Lastly by inhibiting the TLR pathway, we can control the inflammation. But an in-depth further experimental and clinical research is required to develop the novel therapeutic drugs in this area.
Protein Tyrosine Phosphatase (PTP)-1B Inhibitors
Tyrosine phosphorylation is one of the basic mechanism involved in cell growth and differentiation. Insulin upon binding to its receptor activates the insulin receptor tyrosine kinase (IRTK) through auto phosphorylation mechanism. This leads to the activation of recruitment of insulin receptor substrate (IRS)-1, followed by the activation of phosphatidyl inositol 3 kinase (PI3K) and subsequent translocation of glucose transporter-4 (GLUT-4).88In contrast, tyrosine phosphatase enzymes dephosphorylate them, and inactivates the fundamental cellular process like cell differentiation, metabolism, and cell apoptosis.89 Along with this, other PTPs which are involved in negative regulation of the insulin secretion are leukocyte antigen-related tyrosine phosphatase(LAR), SH2-domain-containing phosphotyrosine phosphatase (SHP2), receptor protein tyrosine phosphatase (rPTP), and protein tyrosine phosphatase 1B (PTP1B). Certainly, PTP1B seemed to be a key regulator in insulin receptor signalling90 and inhibition of PTP1B is a novel receptor target in the treatment of DM.
Conclusion
Receptors always play a prominent role in physiology and pathology of various disease conditions in humans as well as in animals. Due to interdependency and complexity of receptors in their mechanism, one can’t corroborate their involvement in the pathophysiological event of the disease. However, we can conclude that receptor-ligand interaction mechanism can be used in development of new therapeutic agents by gaining sufficient knowledge on it, and thereby, designing the new drugs based on ligand-receptor interaction for better optimal drug discovery. The researchers have to develop the alternative chemical moieties that retain its affinity to receptor and efficacy. So, one can utilise these preliminary findings to develop novel therapeutic agents that target the selective receptor providing better therapy for DM. Finally, DM is a metabolic disorder due to the glucose surfeit, it alarms the need to maintain it levels by optimising medications along with regular excercise and changes in diet.
Acknowledgements
Authors would like to say deep gratitude of thanks to Ms. V. Rajani Sekhar for her support in editing the manuscript.
References


